For example, two electron domains gives a linear molecule three domains gives a trigonal planar molecule four domains gives a tetrahedral structure five dom. Electron geometry refers to the geometry of electron domains around the central atom and can be determined from a formula (Niles, 2004). Shape is determined by the number of both shared and unshared domains of electrons around the central atom and refers only to the orientation of atoms in and around the center.Ĥ. The molecular geometry can be determined from the Lewis Dot diagrams because it is determined by the number of electron domains around the central atom (Niles, 2004). The Lewis Dot structure is used to represent the valence electrons in chemical bonding, and the Lewis Dot structures are useful for explaining the chemical bonding in ions and molecules (Chieh, 2004). The molecules are considered isoelectronic because they both have three hydrogen atoms attached to a highly negative atom and have the same electronic potential.ģ. The nitrogen atom has a lone pair of electrons so it acts as a base. The NH3 molecule also has a trigonal pyramidal configuration, giving the molecule an overall dipole moment and making it polar (Ammonia, 2005). The hydronium ion, H3O, has a positive charge on one of the H ions in the molecule, which has a trigonal pyramidal configuration. The molecule is trigonal planar because there are three electron domains around the central sulfur atom.Ģ. 
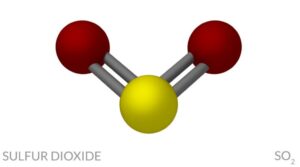
The molecule has an S-O single bond and an S=O double bond. The SO2 molecule is a bent molecule, existing as a resonance structure, with a lone pair of electrons on the S. There is no charge on the molecule because there are no lone electron pairs. There are two electron domains around the central carbon atom, therefore it is linear (Niles, 2004). The molecule has an S-O single bond and an SO. The difference in polarity between CO2 and SO2 can be explained by their molecular shape. The SO2 molecule is a bent molecule, existing as a resonance structure, with a lone pair of electrons on the S. Its molecular geometry and its electron-domain geometry is tetrahedral.1. For example, carbon tetrachloride, CCl4, has no lone pairs on the central carbon atom. If the central atom of a molecule has no lone pairs, the molecular geometry and the electron-domain geometry are the same. The lone pairs are not considered when determining molecular geometry, only the bonds with the atoms are considered. The polarity influences the bond angle for the bent geometry. 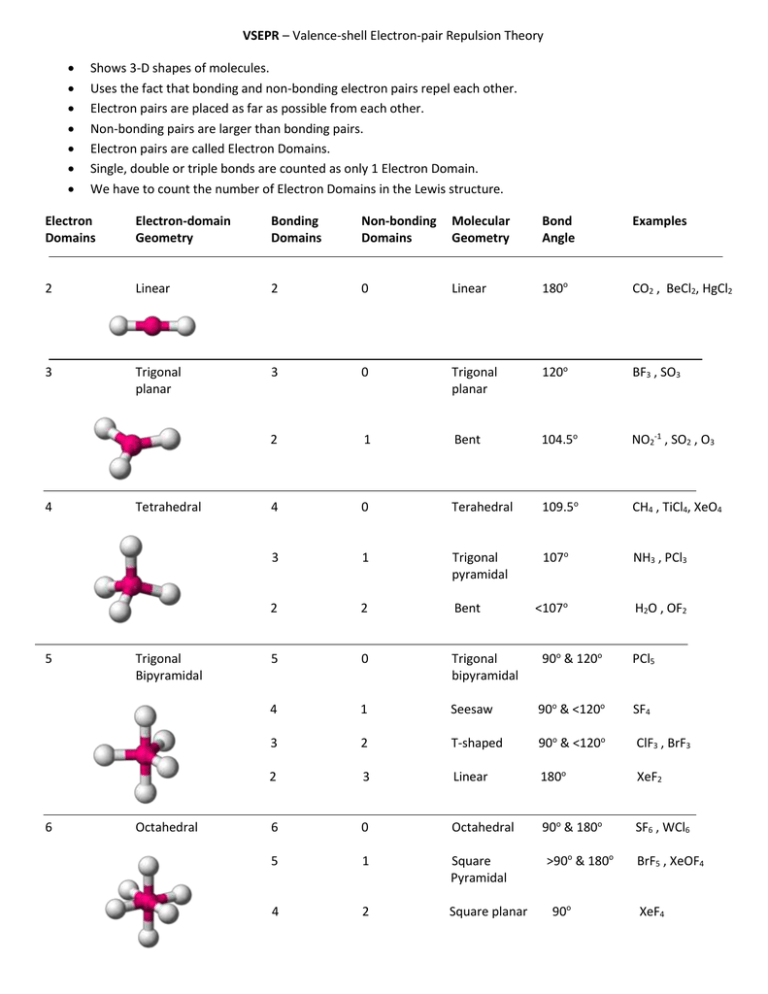
Since the difference in polarity is greater than zero, SO2 is a polar molecule. Note that the bent shape is related to the diffence in electronegativity between sulfur and oxygen (3.5 - 2.5). When referring to the molecular geometry for sulfur dioxide, the molecular geometry is bent. Also related to electron-domain geometry is the fact that the sulfur has sp2 hybridization, since its geometry is trigonal planar. This is because it has 3 electron domains - the 6 valence electrons for sulfur form 2 single bonds with 2 oxygen atoms and sulfur has one non-bonding lone pair. For example, sulfur dioxide, SO2, electron-domain geometry is trigonal planar. A molecule can have a different shape when referring to its electron-domain geometry than when referring to its molecular geometry. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
